Research
The main goal of our research activities is to design and synthesize new RNA ligands bearing both strong affinity and high selectivity for a biologically relevant RNA target and to study the interactions involved in the formation of the ligand-RNA complex using biochemical and biophysical techniques.
- Antimicrobials
-
Therapeutic innovations in antibiotics : focus on the targeting of toxin-antitoxin systems using small-molecule RNA binders
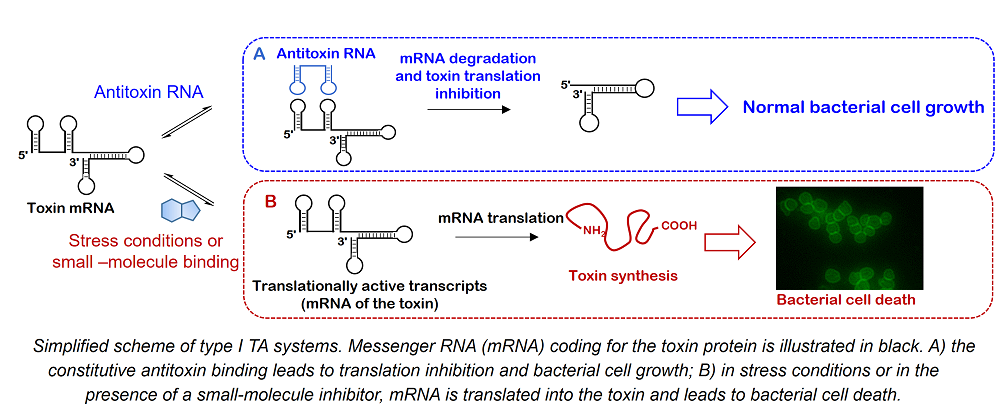
There is currently an urgent need for new antibiotics in order to overcome the steady emergence of multidrug-resistant bacteria and the associated human and economic cost. For the development of new antimicrobial agents, two major issues must be overcome : the difficulty to permeate bacterial membranes and the toxicity of compounds. Furthermore, the available number of specific targets remains restricted. All these issues led to a strong decrease in the efforts done toward the discovery of new antibiotics both in industry and in academia. In this context, the purpose of this project is the discovery of new antibiotics targeting original and so far unexploited targets : bacterial toxin-antitoxin (TA) systems. TA systems are small genetic elements composed of a toxin gene and its cognate antitoxin both coding for corresponding toxin and antitoxin products. The toxins of all known TA systems are proteins able to inhibit bacterial cell growth or lead to cell death, whereas the antitoxins are either proteins or small regulatory RNAs that neutralize the toxin. Here, we decided to target type I TA systems where the antitoxin is a non-coding RNA that binds to the messenger RNA (mRNA) coding for the toxin thus inhibiting its translation.
Innovantibio This project is supported by ANR-PRC grant (ANR-PRC ANR-17-CE18-0009, 2018-2022) and is performed in collaboration with Dr. Fabien Darfeuille at ARNA Laboratory in Bordeaux.
ANR - Anticancer strategies
-
Targeting oncogenic microRNAs: toward new chemotherapies
We have designed and synthesized various new series of RNA ligands able to bind selectively to two precursors of oncogenic microRNAs (pre-miR-372 and pre-miR-373) involved in the proliferation and tumorigenesis of gastric adenocarcinoma. The development of a cell-free high-throughput assay allowed the selection of most promising compounds for intracellular studies. We identified compounds able to selectively inhibit the production of oncogenic miRNAs and inhibit cancer cells proliferation and restor normal mRNA translation.
We also applied the HTS assay to the screening of larger libraries. As an example, the screening of the Essential Library belonging to the French National Chemical library led to the discovery of polyamine derivatives as promising compounds able to interfere with the biogenesis of oncogenic miRNAs.
Treating cancer as an infectious disease with antibiotics
Compelling evidence suggests that cancer stem cells (CSC) are the roots of current shortcomings in advanced and metastatic colorectal cancer treatment. CSC represents a minor subpopulation of tumor cells endowed with self-renewal and multi-lineage differentiation capacity which can escape from both conventional and targeted therapies (cetuximab, avastin), disseminate and seed metastasis. For that reason, Targeting CSC has become a major goal to design new therapeutic routes that may prevent tumor relapse and metastasis. Most drugs possess off-target effects that might provide substantial benefit for cancer treatment. Our recent work suggests that some antibiotics are able to interfere with stem-like properties -such as self-renewal- inherent to CSC phenotype. This project aims to determine whether these compounds can be used as adjuvant during the course of classic chemotherapy to target CSC and prevent disease recurrence and metastatic process.
The project is performed in collaboration with Dr. Alexandre DAVID at Institut de Génomique Fonctionnelle in Montpellier.
This work is supported by INCa grant (INCa PLBio 2017-160, 2018-2020) - Antiviral strategies
-
Nouvel onglet contenu